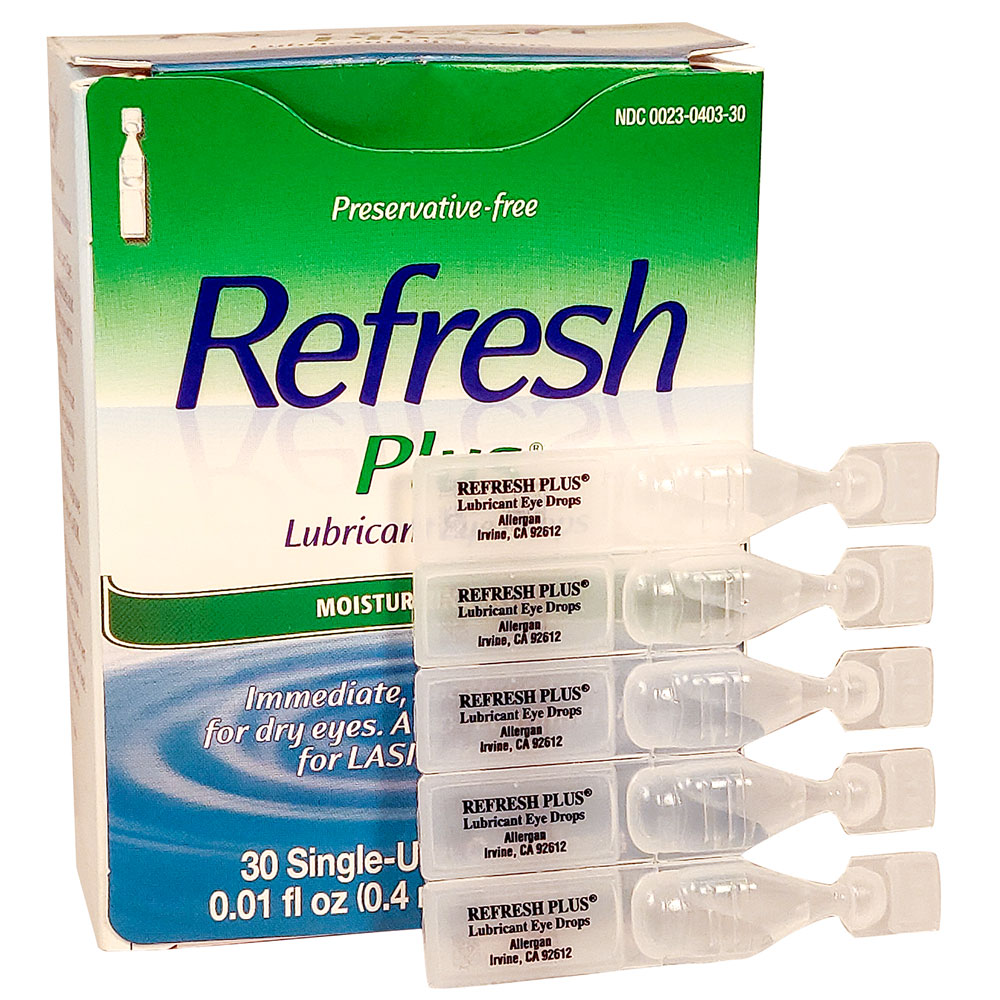
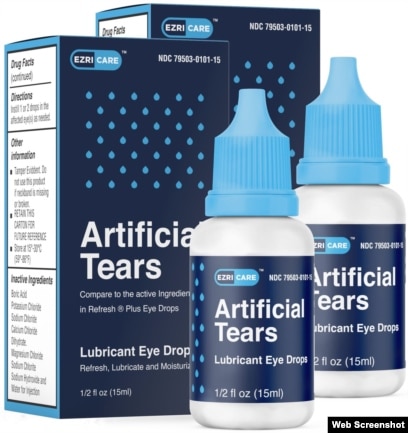
Amazon.com: Refresh Plus Lubricant Eye Drops, Preservative-Free, 0.01 Fl Oz Single-Use Containers, 30 Count (Pack of 1) : Health & Household
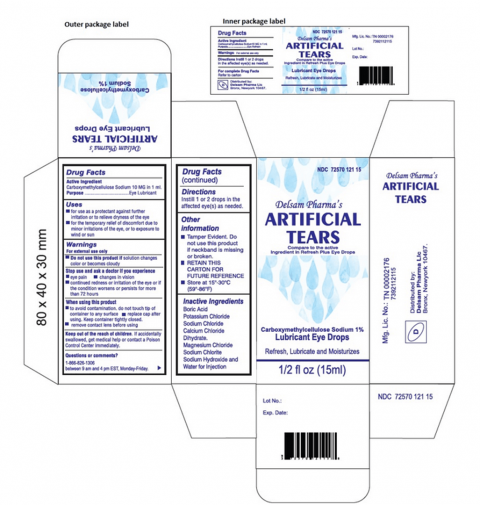
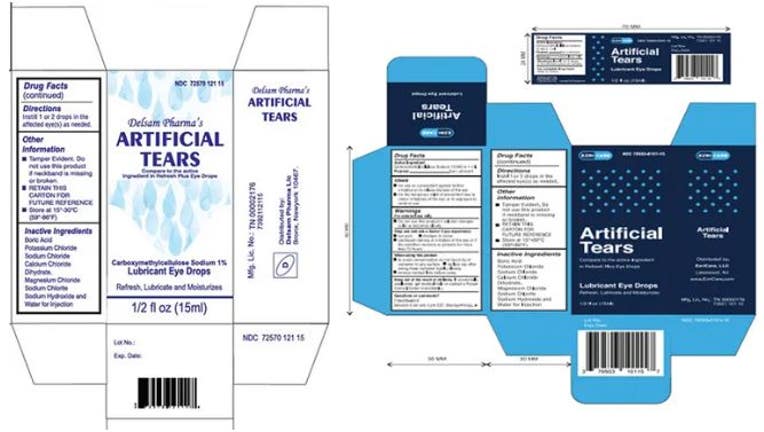
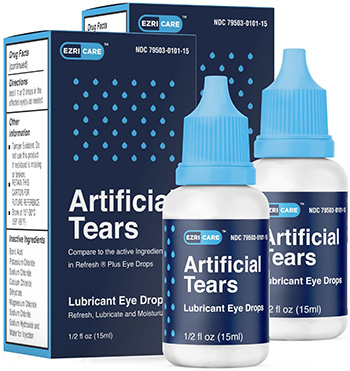
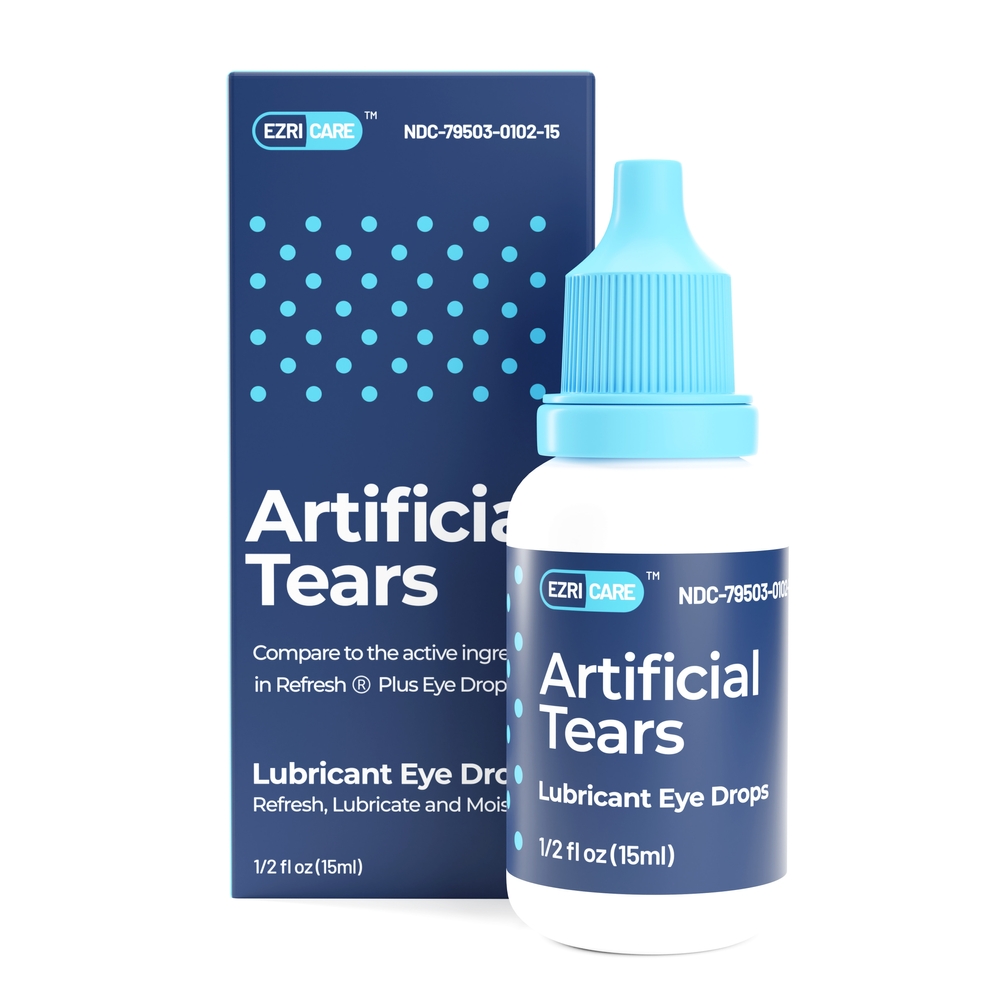
Global Pharma Healthcare Issues Voluntary Nationwide Recall of Artificial Tears Lubricant Eye Drops Due to Possible Contamination | FDA